
Nickel (element symbol Ni, atomic number 28) is a transition metal with a variety of stable physicochemical properties and is a key strategic raw material for modern industry and new energy industries. It is a transition metal element in Group VIII of the fourth period of the periodic table and an indispensable key metallic material in the global industrial system. In traditional applications, nickel is a core raw material for stainless steel production, alloy preparation, and electroplating processes. With the rapid development of the new energy industry, nickel has become a core component of lithium-ion battery cathode materials, and market demand continues to expand. This article, based on the fundamental properties of nickel, systematically elucidates its discovery history, physicochemical characteristics, resource distribution, processing technology and supporting equipment, industrial applications, and market trends, comprehensively covering all dimensions of the industrial utilization of nickel and providing a reference for research and production in related fields.
The discovery history and naming origin of nickel

The English name for nickel, "Nigel," originates from the German word "Kupfernickel." Its common name, "devil's metal," is directly related to mining activities in the 17th century. In the 17th century, during copper mining operations in Saxony, Germany, workers discovered a dark red ore often covered with a green hue. Multiple attempts to extract metallic copper from it failed. Simultaneously, workers who came into contact with and smelted the ore experienced poisoning symptoms, including abdominal pain, vomiting, diarrhea, and dizziness; in severe cases, they suffered from confusion, shock, and even death. The miners believed the ore was copper in disguise, a manifestation of an evil spirit, and named it "Kupfernickel," meaning "Old Nick's copper." "Old Nick" is the name of an evil spirit in Saxon mythology and a local colloquial term for the devil; this is the core origin of nickel's nickname, "devil's metal."
From 1751 to 1754, Swedish chemist Axel Cronstätter conducted systematic experimental studies on the ore, confirming that it was nickel arsenide (red nickel arsenide), and that the arsenic in the ore was the direct cause of the workers' poisoning. During the experiments, researchers found that the chemical reaction characteristics of the ore were completely inconsistent with those of copper compounds. After reducing the ore with charcoal, a hard, silvery-white metal was obtained, whose appearance, magnetism, and other physical characteristics differed significantly from copper. This confirmed the isolation of a completely new metallic element. This new element was named Nickel, after its raw material, "Kupfernickel."
Core physicochemical properties of nickel

The basic atomic parameters and physicochemical properties of nickel are the core basis for its industrial application. Its key parameters and properties are shown in the table below.
| Parameter Category | Specific Indicators |
| Element Symbol | Ni |
| Atomic Number | 28 |
| Relative Atomic Mass | 58.69 |
| Crystal Structure | Face-centered cubic (FCC) |
| Melting Point | 1453°C |
| Boiling Point | 2732°C |
| Density at Room Temperature | 8.908 g/cm³ |
| Mohs Hardness | 4 |
| Ductility | High ductility, can be cold-rolled processed |
| Magnetic Properties | Ferromagnetic at room temperature |
| Chemical Properties | Resistant to corrosion and oxidation at room temperature, can form alloys with various metals, possesses catalytic properties, can achieve coating deposition via electrolysis |
| Recycling Characteristics | Fully recyclable, no performance degradation after multiple recycling cycles |
Based on the above physicochemical properties, and addressing high-frequency issues in industrial applications, the following objective conclusions are drawn:
- Corrosion Resistance: Nickel and nickel-based alloys possess corrosion resistance. Pure nickel can withstand high temperatures, various chemical media, and high humidity environments. Nickel-plated steel only exhibits corrosion resistance, not complete corrosion protection. When nickel is exposed to extreme environments for extended periods, a nickel oxide layer forms on its surface, leading to corrosion. Based on its corrosion resistance, nickel can be used to prepare various alloy materials such as forged nickel, nickel-iron, nickel-chromium, and nickel-copper.
- Magnetic Properties: Nickel is one of only four metals possessing ferromagnetism at room temperature; the other three are iron, cobalt, and gadolinium. Nickel can be attracted by magnetic fields and can generate its own magnetism, although its magnetic strength is lower than that of iron and cobalt. Alloy materials made from aluminum, nickel, and cobalt can be made into permanent magnets that retain their magnetism even after being heated to a red-hot state.
- Nickel Plating Process: Nickel plating is a surface treatment process that deposits a nickel layer on the surface of a substrate. It can alter the substrate's conductivity, hardness, adhesion, corrosion resistance, wear resistance, and appearance. Nickel plating processes are divided into two categories: electroplating, which deposits a nickel layer on the substrate surface through an electrolytic reaction; and electroless plating, which completes the nickel layer deposition through an autocatalytic chemical reaction, requiring no external power source.
- Recycling Performance: Nickel is fully recyclable, and its physicochemical properties do not degrade after multiple recycling cycles, making it one of the most recyclable metal materials globally. Statistics show that approximately 68% of nickel globally can be recycled from consumer products, primarily from waste stainless steel and used batteries. Nickel recycling can promote the development of a circular economy and alleviate resource supply gaps caused by expanding demand.
Nickel ore resource types and global distribution characteristics
Industrial types of nickel ore
Nickel ores with economic mining value are divided into two main categories: sulfide nickel ores and laterite (oxidation) nickel ores. Laterite nickel ores can be further divided into two subtypes: limonite type and sapropel type. The characteristics and industrial applications of different types of nickel ores are clearly different.
- Sulfide nickel ore: This type of deposit is a traditional source of high-purity nickel, producing Class 1 nickel with a nickel content of not less than 99.98%, a core raw material for the production of nickel sulfate for batteries. Currently, global reserves of high-quality, large-scale sulfide nickel ore are continuously decreasing, and the difficulty of exploring new deposits is constantly increasing.
- Limonite-type laterite nickel ore: This type of deposit is relatively easy to mine, but the nickel grade of the ore is low. It requires high-pressure acid leaching (HPAL) to produce Class 1 nickel. Only a portion of this type of ore is suitable for the production requirements of battery-grade high-purity nickel.
- Saprophytic laterite nickel ore: This type of ore can be processed to produce Class 2 nickel, which cannot meet the production requirements of battery-grade high-purity nickel. It mainly supplies the stainless steel production industry.
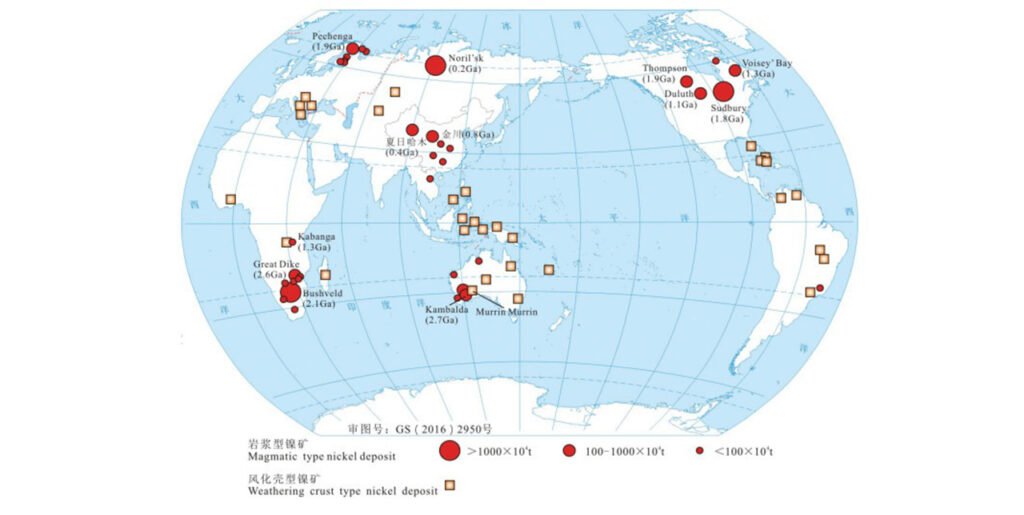
Global Nickel Ore Resource Distribution and Supply Pattern
Global nickel ore resources exhibit a significant geographical concentration. Economically viable nickel deposits are primarily located in Australia, Indonesia, South Africa, Russia, and Canada, which together account for over 50% of global nickel reserves. Lateritic nickel ore is mainly distributed near the equator, while sulfide nickel ore is primarily found in South Africa, Russia, and Canada. Specific reserve distributions are shown in the table below.
| Deposit Type | Country / Region | Reserves (million tonnes) | Global Total for Same Deposit Type (million tonnes) |
| Laterite nickel deposits | Indonesia | 3329.1 | 17815.25 |
| Laterite nickel deposits | Australia | 3152.9 | 17815.25 |
| Laterite nickel deposits | Philippines | 1801.34 | 17815.25 |
| Laterite nickel deposits | Cuba | 1617.4 | 17815.25 |
| Laterite nickel deposits | New Caledonia | 1501.6 | 17815.25 |
| Laterite nickel deposits | Brazil | 1482.42 | 17815.25 |
| Sulfide nickel deposits | South Africa | 3321.63 | 11803.76 |
| Sulfide nickel deposits | Canada | 2192.31 | 11803.76 |
| Sulfide nickel deposits | Russia | 2045.75 | 11803.76 |
| Sulfide nickel deposits | Australia | 1187.85 | 11803.76 |
Regarding the global nickel ore supply structure, more than 25 countries currently engage in nickel mining operations. The Asia-Pacific region contributes 65% of global nickel production, with Indonesia, the Philippines, Russia, Australia, and Canada being the world's top five nickel producers, exhibiting a diversified supply chain. Furthermore, there are nickel deposits with development potential in offshore areas, which are expected to achieve commercial development in the future as mining technologies and regulatory systems improve.
Nickel ore beneficiation and processing technology and supporting core equipment
Different types of nickel ore require different beneficiation and smelting processes. Each process stage requires specialized equipment to complete continuous operation. The selection of equipment, operating parameters, and process adaptability directly determine processing efficiency, metal recovery rate, and final product quality. The following details the working principles, process functions, and operating requirements of the equipment throughout the entire process, categorized by nickel ore type and corresponding process.
Complete equipment for the beneficiation and smelting of sulfide nickel ore
The mainstream processing route for sulfide nickel ore is crushing and screening - washing - grinding and classification - flotation enrichment - pyrometallurgical smelting - electrolytic refining, ultimately producing Class 1 high-purity electrolytic nickel. The supporting equipment for each step is as follows:
Crushing, screening and washing equipment
The core objective of this step is to crush the large blocks of raw ore to the target particle size, while removing clay impurities from the ore to prevent mud formation in subsequent processes.
- Jaw Crusher: Used for the initial coarse crushing of raw ore. The core structure consists of a fixed jaw plate and a moving jaw plate. Its working principle is that the moving jaw plate reciprocates periodically relative to the fixed jaw plate, applying compression and splitting forces to the ore. It can crush raw ore with a maximum particle size of over 1000mm to medium-sized ore of 100-300mm, meeting the feed requirements of subsequent medium and fine crushing processes.
- Cone Crusher: Used for medium and fine crushing of ore. It comes in two types: standard and short-head. The standard type is used for medium crushing, and the short-head type for fine crushing. Its working principle is that the moving cone oscillates within the fixed cone cavity, continuously compressing and grinding the ore. It can crush the coarsely crushed ore to a particle size of 12-25mm, meeting the feed particle size requirements of the grinding process.
- Linear Vibrating Screen: Installed at the discharge end of the crushing process, forming a closed-loop circulation with the fine crushing equipment. The working principle is to generate high-frequency linear vibration through a vibrator, causing the ore to be classified according to the screen aperture size on the screen surface. Ore with the required particle size enters the next process, while oversized ore is returned to the fine crushing equipment for further crushing, ensuring uniform particle size of the ore entering the mill.
- Drum washing pulper machine: Used for pretreatment of nickel sulfide ore with high mud content. The working principle is to remove clay impurities adhering to the ore surface through the rotation of the drum and high-pressure water washing, preventing muddy minerals from affecting the subsequent flotation separation effect.
Grinding and classifying equipment

The core objective of this step is to achieve the individual dissociation of nickel sulfide minerals and gangue minerals, while controlling the particle size of the grinding product and reducing over-grinding.
- Rod mill: Primarily used for single-stage grinding operations. The cylinder is filled with steel rods as grinding media. The working principle is that the steel rods rotate with the cylinder, crushing the ore through line contact impact and grinding. The ground product has a uniform particle size, minimal over-grinding, and avoids the over-grinding and mudding of nickel sulfide minerals.
- Overflow ball mill: Primarily used for two-stage grinding operations. The cylinder is filled with steel balls of different sizes as grinding media. The working principle is that the centrifugal force and gravity generated by the rotation of the cylinder cause the steel balls to fall and cascade, impacting and grinding the ore to the target fineness, completely liberating nickel sulfide minerals from gangue minerals. The grinding fineness of nickel sulfide ore is typically controlled at -200 mesh, with 60%-80% of the particles being finer. Specific parameters are adjusted according to the mineral embedding particle size.
- Spiral classifier: Forms a closed-loop cycle with the primary grinding unit. Its working principle involves gravity classification of the ground slurry within a water tank through the rotation of the spiral blades. Coarse particles settle to the bottom and are conveyed back to the grinding mill for further grinding, while fine particles overflow and enter subsequent processes, controlling the upper limit of the particle size of the ground product.
- Hydrocyclone: Used for fine classification in the secondary grinding stage. Its working principle utilizes the centrifugal force generated by the high-speed rotation of the slurry to achieve rapid separation of coarse and fine particles. Its classification efficiency is higher than that of the spiral classifier, allowing for precise control of the particle size distribution of the ground product and improving the separation efficiency of subsequent flotation processes.
Flotation enrichment equipment

The core objective of this step is to enrich nickel sulfide minerals and remove gangue minerals from the milled slurry through flotation separation, thereby obtaining nickel concentrate that meets smelting requirements.
- Mechanically agitated flotation machine: the core equipment for nickel sulfide ore flotation. The volume of a single cell can be adjusted according to the throughput, and multiple cells can be connected in series to form a complete system of roughing, scavenging, and cleaning. The working principle is that the high-speed rotation of the impeller generates negative pressure within the cell, drawing in air and dispersing it into tiny bubbles. Simultaneously, the agitation of the slurry allows the flotation reagents to fully interact with the mineral particles. Hydrophobic nickel sulfide mineral particles adhere to the surface of the bubbles and float to the surface of the slurry, forming a foam layer, which is then scraped off by a scraper to obtain nickel concentrate. Hydrophilic gangue minerals remain in the cell and are discharged as tailings. The roughing section is used for the initial recovery of nickel minerals, the scavenging section is used to improve the overall recovery rate, and the cleaning section is used to improve the grade of the nickel concentrate, ultimately yielding nickel concentrate with a nickel grade of 10%-20%.
Pyrometallurgical equipment

The core objective of this step is to remove impurities such as sulfur, iron, and silicon from the flotation nickel concentrate through high-temperature smelting and blowing to obtain high-nickel matte and crude nickel.
- Rotary drying kiln: Used for drying and dewatering flotation nickel concentrate. The kiln body is installed at an incline and can rotate at low speed. The working principle is that the wet concentrate enters from the high end of the kiln and comes into countercurrent contact with the high-temperature flue gas introduced at the kiln tail, removing free water from the concentrate and reducing the moisture content to below 0.3%, avoiding furnace temperature fluctuations and safety hazards caused by moisture vaporization during smelting.
- Fluidized bed roaster: Used for oxidative roasting of nickel concentrate. The working principle is that air is introduced from the bottom of the furnace by a high-pressure blower, causing the concentrate particles in the furnace to enter a fluidized bed state, fully contacting the air to undergo an oxidation reaction, removing most of the sulfur and arsenic impurities from the concentrate, and simultaneously converting some sulfides into oxides, providing conditions for subsequent smelting processes.
- Submerged arc furnace: Used for matte smelting of roasted concentrate. The working principle is that electrical energy is converted into heat energy through graphite electrodes, raising the temperature of the material in the furnace to 1350-1450℃, causing a melting reaction, and separating into low-nickel matte and slag. Low-nickel matte, with its high density, settles at the bottom of the furnace and is periodically discharged; the slag, with its low density, is continuously discharged from the slag outlet, achieving initial nickel enrichment. The resulting low-nickel matte has a nickel content of 10%-25%.
- Horizontal side-blown converter: Used for the blowing operation of low-nickel matte. Its working principle involves blowing high-pressure air into the molten low-nickel matte through tuyeres, while simultaneously adding quartz flux. This causes impurities such as iron and sulfur to undergo an oxidation reaction, forming slag which is then discharged, yielding high-nickel matte. High-nickel matte can have a nickel content of over 70%, while also enriching valuable metals such as copper and cobalt.
- Refining reverberatory furnace: Used for the roasting and reduction of high-nickel matte, removing residual sulfur and impurities to obtain crude nickel with a nickel content of over 95%, providing raw materials for subsequent electrolytic refining.
Electrolytic refining equipment
The core objective of this step is to purify crude nickel to over 99.98% to obtain high-purity electrolytic nickel that meets the Class 1 nickel standard.
- Electrolytic Cell: The core equipment in the electrolysis process, it is a rectangular tank made of acid-resistant material, internally divided into an anode chamber and a cathode chamber. The anode is made of crude nickel cast anode plate, and the cathode is made of pure nickel starting sheet or stainless steel cathode plate. The electrolyte is a mixed aqueous solution of nickel sulfate and sulfuric acid.
- Rectifying Power Supply: Provides a stable direct current to the electrolysis system. Under the action of the direct current electric field, the crude nickel at the anode undergoes an oxidation reaction and dissolves into nickel ions, which enter the electrolyte. The nickel ions undergo a reduction reaction on the cathode surface, depositing as high-purity electrolytic nickel. Impurities either enter the anode sludge or remain in the electrolyte and are removed through purification.
- Electrolyte Circulation and Purification Device: Used to maintain the stability of the electrolyte composition, remove impurities such as iron, copper, and cobalt from the electrolyte, and ensure the purity of the nickel deposited at the cathode.
- Anode Sludge Treatment Equipment: Used to recover precious metals such as gold, silver, and platinum group metals enriched in the anode sludge, achieving comprehensive resource utilization.
Supporting equipment for hydrometallurgical processing of lateritic nickel ore (HPAL process)
High-pressure acid leaching (HPAL) is the mainstream process for preparing Class 1 battery-grade high-purity nickel from limonite-type laterite nickel ore. The core process is as follows: crushing - grinding and pulping - pre-desiliconization - high-pressure acid leaching - flash cooling - countercurrent washing and solid-liquid separation - solution purification - solvent extraction - product preparation. The supporting equipment for each step is as follows:
Crushing, grinding and pulping equipment

Lateritic nickel ore of the limonite type has high water content and high viscosity. For coarse crushing, a jaw crusher is used, while for medium and fine crushing, an impact crusher is used. The working principle is that the ore is crushed by impact from high-speed rotating hammers, with repeated impacts between the hammers and the impact plates, preventing clogging by sticky ore and crushing the ore to a particle size of 10-20mm.
Grinding is performed using a wet overflow ball mill. The working principle is that the ore, water, and dispersant enter the mill simultaneously, and after grinding by steel balls, a slurry is prepared. The grinding fineness is controlled to be above -200 mesh, with over 80% of the particles, ensuring sufficient liberation of the nickel minerals to meet the requirements of subsequent acid leaching. A hydrocyclone is used for slurry classification, and coarse particles are returned to the ball mill for further grinding.
Pre-desiliconization equipment
The core equipment is an atmospheric pressure stirred leaching tank, equipped with a heating jacket and a stirring device. Its working principle involves thoroughly mixing the slurry and modifier within the tank, reacting under atmospheric pressure and at 80-90℃ to remove active silica from the slurry, thus avoiding scaling problems caused by excessive silica dissolution during high-pressure acid leaching. The stirring device employs a propeller or turbine structure to ensure uniform mixing of the slurry without sedimentation.
High-pressure acid leaching core equipment
The horizontal high-pressure reactor is the core equipment of the HPAL process. Made of titanium or titanium-lined composite steel, it can withstand high-temperature, high-pressure, and highly corrosive environments. The reactor is internally divided into multiple compartments, each equipped with a stirring device. The working principle involves continuously feeding a mixture of mineral slurry and concentrated sulfuric acid into the reactor. Steam heating maintains the reactor at 240-270℃ and 4-5 MPa. Under these high-temperature, high-pressure, and acidic conditions, valuable metals such as nickel and cobalt in the slurry react with the sulfuric acid and dissolve into the liquid phase, while impurities such as iron and aluminum hydrolyze and precipitate, remaining in the solid phase. The residence time of the material in the reactor is controlled to be 60-90 minutes, achieving a nickel leaching rate of over 95%.
Flash cooling and solid-liquid separation equipment
Flash Tank: Used for cooling and depressurizing the slurry after high-pressure acid leaching. It employs a multi-stage series structure, with each stage progressively lowering the pressure. The slurry enters the flash tank after being discharged from the high-pressure reactor, where water rapidly vaporizes, carrying away heat and achieving cooling and depressurization. Simultaneously, the flash steam can be recovered for slurry preheating, reducing system energy consumption.
High-Efficiency Thickener: Used for countercurrent washing (CCD) of the leaching slurry. It also employs a multi-stage series structure, with each thickener equipped with a rake-type agitator. Countercurrent washing fully recovers nickel ions from the leachate, achieving a wash water recovery rate of over 99%.
Horizontal Belt Vacuum Filter: Used for fine filtration of the leaching slurry, yielding a nickel-containing leachate (precious solution) and leaching residue. The leaching residue is washed and then stored in a harmless manner.
Solution purification and solvent extraction equipment
Neutralization Reaction Tank: Used for neutralizing and removing impurities from the leachate. The tank is equipped with a stirring device and an online pH monitoring device. The pH value of the solution is adjusted by adding limestone or lime slurry, causing impurities such as iron, aluminum, and chromium to hydrolyze and precipitate, achieving preliminary purification.
Box-type Mixing and Clarifying Tank: The core equipment for nickel-cobalt separation and solution purification. A single-stage unit consists of a mixing chamber and a clarification chamber; multiple stages are connected in series to form a complete extraction-washing-back-extraction system. The working principle is that the organic phase and aqueous phase are thoroughly contacted by stirring in the mixing chamber. Target metal ions are selectively extracted into the organic phase, while impurities remain in the aqueous phase. Subsequently, the organic and aqueous phases are separated in the clarification chamber. After multi-stage treatment, a high-purity nickel sulfate solution meeting battery-grade requirements can be obtained, with a nickel recovery rate of over 98%.
Product preparation equipment
Evaporator/Crystallizer: Used for evaporating, concentrating, cooling, and crystallizing high-purity nickel sulfate solution to prepare battery-grade nickel sulfate crystals.
Centrifuge and Dryer: Used for solid-liquid separation and drying of nickel sulfate crystals to obtain finished battery-grade nickel sulfate.
Electrolytic Cell: Can electrolyze high-purity nickel sulfate solution to prepare high-purity electrolytic nickel plates with a purity of over 99.99%, meeting Class 1 nickel standards.
Supporting equipment for pyrometallurgical smelting of laterite nickel ore (RKEF process)
The RKEF process is the mainstream process for producing nickel-iron alloys from sapropelic laterite nickel ore. The product is Type 2 nickel, mainly supplied to the stainless steel industry. The core process is crushing, drying, rotary kiln pre-reduction, electric arc furnace smelting, and refining. The supporting equipment for each stage is as follows:
Crushing and Drying Equipment
Jaw crushers and impact crushers are used to crush the raw ore to a particle size of 10-30mm. The moisture content of sapropelic laterite nickel ore is typically 30%-40%. A rotary kiln is used for drying and dehydration. The kiln is 30-60m long, installed at an incline, and rotates at low speed. High-temperature flue gas comes into counter-current contact with the ore to remove free water, reducing the moisture content to below 0.5%, thus preventing moisture from affecting furnace temperature stability during the pre-reduction process.
Pre-reduction Equipment

The rotary kiln is the core pre-reduction equipment in the RKEF process. The kiln is 60-120m long and 3-6m in diameter, installed at an incline, and the operating temperature is controlled at 800-1000℃. The working principle is as follows: dried ore is mixed with reducing agent and flux in a specific ratio and fed into the kiln from the tail. As the kiln rotates, it moves towards the kiln head, where it comes into counter-current contact with the high-temperature flue gas generated by combustion. Nickel and some iron oxides in the ore are reduced to a metallic state, yielding pre-reduced roasted ore with a metallization rate of 20%-30%. The hot roasted ore discharged from the kiln head is directly fed into the electric arc furnace (EAF) to reduce heat loss.
Smelting Equipment
A closed-type EAF is used for the melting and reduction of pre-reduced roasted ore. The working principle involves converting electrical energy into heat energy through graphite electrodes. The furnace temperature is maintained at 1500-1600℃. The roasted ore undergoes a melting reaction inside the furnace, separating into liquid nickel-iron alloy and slag. The nickel-iron alloy settles at the bottom of the furnace and is periodically discharged from the taphole; the slag is continuously discharged from the slag outlet. The furnace has a closed structure and can recover carbon monoxide gas generated inside the furnace as fuel for the rotary kiln, reducing system energy consumption. The produced nickel-iron alloy has a nickel content of 8%-15%.
Refining Equipment
The ladle refining furnace is used for refining liquid nickel-iron alloys. By adding refining agents, impurities such as sulfur, phosphorus, and carbon are removed from the alloy, and the alloy composition is adjusted to obtain a finished nickel-iron alloy that meets the requirements for stainless steel production.
Industrial applications of nickel

Thanks to its stable physicochemical properties, nickel has applications in multiple fields, including metallurgy, new energy, surface treatment, materials preparation, and environmental protection. Its core applications are as follows:
- Stainless Steel and Alloy Production: Stainless steel is a traditional core application area for nickel. Nickel enhances the corrosion resistance, ductility, and weldability of stainless steel, and is the core alloying element of austenitic stainless steel. Simultaneously, nickel can form alloys with various metals such as iron, chromium, copper, and aluminum to prepare high-temperature resistant, corrosion-resistant, and high-strength special alloys, widely used in aerospace, shipbuilding, chemical equipment, bridge construction, and other fields.
- New Energy Battery Field: Nickel is a core component of lithium-ion battery cathode materials. It can improve battery energy density and storage capacity while controlling production costs, making it a core raw material for high-nickel ternary cathode materials. With the expansion of the global electric vehicle market, the battery sector has become the fastest-growing application area for nickel demand.
- Surface Treatment Field: Nickel plating processes can form a uniform nickel coating on the surface of metallic and non-metallic substrates, improving the substrate's corrosion resistance, wear resistance, and surface hardness, while also improving the substrate's appearance. It is widely used in hardware products, electronic components, mechanical parts, automotive parts, and other fields.
- In the field of magnetic materials: Leveraging its room-temperature ferromagnetic properties, nickel can be used to prepare permanent magnet materials. AlNiCo permanent magnets, in particular, exhibit magnetic stability at high temperatures and are widely used in motors, instruments, sensors, and other fields.
- In the field of catalytic materials: Nickel possesses stable catalytic properties and can be used as a catalyst for chemical reactions such as hydrogenation, dehydrogenation, and reforming. It is widely used in petrochemicals, organic synthesis, and coal chemical industries.
- In the field of agriculture and environmental protection: Nickel is a trace element essential for plant growth and can be used to prepare trace element fertilizers. Simultaneously, nickel-based catalysts can be applied to the purification of industrial wastewater and waste gas, achieving the degradation and transformation of pollutants.
Conclusion
Nickel is a key strategic metal in modern industrial systems and new energy industries. Its name originates directly from early mining activities, and it has been confirmed as an independent metallic element through systematic experimental research. Nickel possesses stable physicochemical properties such as a high melting point, corrosion resistance, room-temperature ferromagnetism, easy alloying, and complete recyclability, making it a core raw material for industrial production in many fields. Global nickel ore resources are divided into two main categories: sulfide-type and laterite-type. Laterite-type nickel ore is further divided into limonite-type and sapropelic-type deposits, with different types of deposits suited to different processing technologies and applications. Global nickel ore resources exhibit a significant geographical concentration, with the Asia-Pacific region being the core area for global nickel production. Sulfide-type nickel ore is the traditional source of high-purity Type 1 nickel, but its reserves are continuously decreasing, making laterite-type nickel ore the core direction for future nickel resource development. Different types of nickel ore require differentiated beneficiation and smelting processes. Sulfide nickel ore uses a flotation-pyrometallurgical-electrolytic refining process, limonite-type laterite nickel ore uses the HPAL hydrometallurgical process, and sapropelic laterite nickel ore uses the RKEF pyrometallurgical process. Each process requires specialized equipment for crushing, grinding, flotation, smelting, leaching, extraction, and electrolysis. The selection of equipment, its operating parameters, and its compatibility with the process directly determine processing efficiency, metal recovery rate, and final product quality. Nickel has applications in stainless steel and alloy production, new energy batteries, surface treatment, magnetic materials, and catalytic materials, among others. The new energy battery sector has become the core driver of nickel demand growth, propelling the continued expansion of global nickel demand.


















